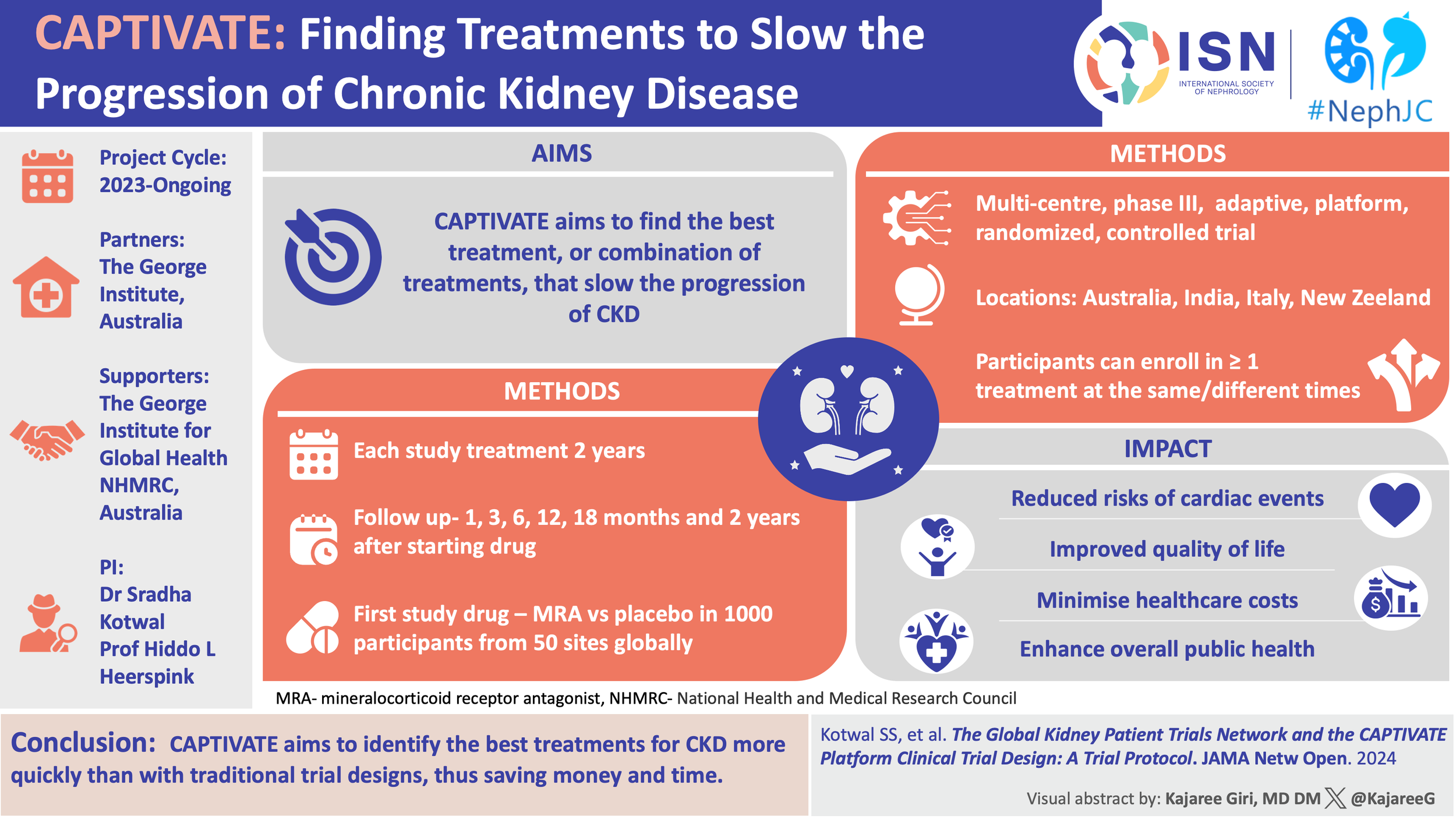
#NephTrials chat
Tuesday February 16th 9 pm Eastern
Introduction
The year 2020 was the year of the COVID-19 pandemic and has challenged health care and health care providers around the globe in a myriad of ways. The challenges we faced were not only in regards to understanding the virus and developing effective treatments against it but also in implementing the findings into practice; at times ineffective treatments (found effective in poorly designed trials) being delivered widely.
…evidence based practice, needs practice based evidence…
One of the most consistent findings in health services research is the gap between best practice as suggested by scientific evidence and its actual implementation and delivery to patients. Thus evidence based practice, needs practice based evidence and the research findings produced must be appropriately implemented and made relevant to the patients who need it the most.
How can we achieve this goal? Can this be achieved by doing clinical trials in a more natural environment thereby making the research findings rapidly translatable to clinical practice?
In this context, pragmatism in clinical trials arose from concerns that many trials did not adequately inform practice because they were optimized to determine efficacy.
What is the difference between efficacy and effectiveness?
Efficacy is the impact a treatment exerts under optimal circumstances, typically maintained in a traditional RCT.
Effectiveness refers to the impact of treatment under usual treatment conditions, in which patient, provider and service system factors affecting treatment outcomes are not controlled.
There is often a gap between efficacy and effectiveness which hampers translation of research findings and new treatments into routine care.
Traditional RCT designs are naturally geared towards internal validity- typically designed with strict inclusion-exclusion criteria, ensure strict protocol adherence in the optimal clinical setting and are usually done in high volume centers with the most experience in the clinical setting that is being studied. External validity needs to be consciously designed into the trial to maintain generalizability. Without external validity, the findings of the trial may not be applicable to the world outside of the trial setting.
Enter the need for pragmatic trial designs!
How did pragmatic trials arise?
Wikipedia defines pragmatism as
Pragmatism is a philosophical tradition that considers words and thought as tools and instruments for prediction, problem solving, and action, and rejects the idea that the function of thought is to describe, represent, or mirror reality. Pragmatists contend that most philosophical topics—such as the nature of knowledge, language, concepts, meaning, belief, and science—are all best viewed in terms of their practical uses and successes.
Charles Peirce, William James and Thomas Dewey are credited, around the 1870s, with conceptualizing and popularizing this term. But what does that have to do with pragmatic trials? Jump forward a century. In 1967, Schwartz and Lellouch described RCTs as being either pragmatic or explanatory (Schwartz et al J Chr Dis1967). They coined the terms “pragmatic” to describe trials that help users choose between options for care, and “explanatory” to describe trials that test causal research hypotheses (i.e., that a given intervention causes a particular benefit).
Why should we do pragmatic trials and how are such trials unique?
Pragmatic trials are designed with the intention to inform a clinical or policy decision by providing evidence for adoption of the intervention into real-world clinical practice.
The optimal effect of an efficacious new treatment can be distorted by real world factors. And hence we need pragmatic trials for certain interventions to make important policy decisions or to drive change. A good example for this was elaborated here on the non-pharmacological interventions to reduce BP (Hiremath, J Clin Hyp 2018). The DASH trial was an efficacy trial which showed that a low sodium diet, achieved in the trial by meal provision, improved BP control. But such an intervention of providing all the meals including snacks and cooked meals is not feasible in real life. Not surprisingly, a subsequent lifestyle intervention trial with only counselling without meal provision, did not achieve the same effect on lowering sodium in diets of the participants.
There are many unique features and specific limitations of pragmatic trials:
Unlike the strict selectivity and inclusion criteria of a traditional RCT, pragmatic trials are designed so that they incorporate the variations seen between patients in routine clinical practice and are usually designed to include participants reflecting those seen in clinical practice to whom the treatment interventions will be applied. The inclusion criteria are typically broad and participants are quite heterogeneous with respect to their characteristics.
Treatment may be tailored to patient needs and is kept flexible with respect to delivery and adherence, to mirror routine practice. This can be a major limitation as it can obliterate our ability to actually see a difference in the two arms of intervention vs control.
Follow up in such trials is typically based in a usual care setting attempting not to put extra burden on patients.
The primary outcome studied typically focuses on patient related outcomes and priorities important to patients.
Open label design - reporting of adverse events, reasons for treatment discontinuation, and many patient reported outcomes are subject to greater degrees of bias in open trials and can affect trial quality
The problem with informed consent: Can it affect the pragmatic nature? Often, the inherent design of the pragmatic trial makes obtaining individual informed consent impractical, especially in cluster RCT designs and can raise ethical issues.
Are pragmatic trials the same as cluster randomized trials?
Most, but not all cluster randomized trials are pragmatic RCTs. Clusters can constitute clinics, schools, villages etc. These trial designs are used when individual randomization is either logistically impractical or not feasible. Cluster randomization which involves groups of patients ( as opposed to individual patients) who are randomly assigned to the same intervention is popular in pragmatic trials. A pragmatic approach is easier when an intervention is implemented at a group level rather than at an individual level — this is one reason that pragmatic trials commonly incorporate cluster randomization rather than individual randomization. Watch out for another edition of #NephTrials for a discussion of Cluster RCTs.
However, a pragmatic trial need not always be a cluster randomized. Even an individual patient level RCT can be pragmatic.
Are there any criteria or guidelines for what makes a pragmatic trial?
Exactly what makes a trial pragmatic? Rather than a clear dichotomization, there are various domains, and trials could have some domains which are more pragmatic than others. This lead to the formation of the PRECIS guidelines (Thorpe et al, J Clin Epi 2009), which were revised a few years later.
Figure 1 from Thorpe et al, J Clin Epi 2009
Some examples worked out are shown below:
Figure 2 from Thorpe et al, J Clin Epi 2009
A few years later, the PRECIS-2 guidelines (Loudon et al, BMJ 2015) made a few more changes, and you can see the modified domains below
The PRECIS-2 Toolkit is available here: https://www.precis-2.org/
The NIH also has provided useful guidance in this document https://rethinkingclinicaltrials.org/chapters/pragmatic-clinical-trial/what-is-a-pragmatic-clinical-trial/
What are examples of pragmatic and non pragmatic trials in the past?
An excellent recent example of Cluster RCTs are the two studies which compared the use of isotonic saline with that of balanced crystalloid solutions- SALTED and SMART. See our coverage on these two trials at #NephJC and also the accompanying #NephStats commentary on pragmatic trials.
There have been some completed and ongoing pragmatic trials in dialysis - most of these below are all cluster RCTs:
Why do we need Pragmatic trials in dialysis patients?
A meta-analysis (Smyth et al, JAMA IM 2019) of 189 trials aiming to assess the generalizability of large, multicenter RCTs among patients undergoing dialysis found that trial participants were significantly younger, with differing pattern of comorbidities and much lower mortality rates than patients in the largest US national registry of dialysis patients. If trial participants are not like dialysis patients we see everyday, can we extrapolate the evidence so easily? Thus more pragmatic trials to effectively and meaningfully test the interventions in the dialysis patient population is the logical next step.
Specifically, the dialysis units are well suited for pragmatic RCTs because of standardized approaches to treatment and data acquisition, and a well-developed infrastructure for delivering care to patients.
In this edition of #Nephtrials, we will be discussing the PHOSPHATE trial, as an example of a pragmatic trial.
What do we know so far about bone mineral disease, calcification and kidney disease?
Pathophysiology of secondary hyperparathyroidism is quite complicated and leads to rise in PTH and serum phosphorus with progressive loss of GFR. Basic science research has shown high phosphate to induce phenotypic changes in vascular smooth muscles which leads to increased predisposition to vascular calcification (Jono et al, Circ Res 2000). But do mechanistic effects of drugs studied in the lab or in animal models always translate into clinically meaningful effects in actual patients?
What kind of trials have we seen in the CKD-MBD area?
Observational studies (eg Block et al, JASN 2004) in dialysis patients have shown an association between hyperphosphatemia and hyperparathyroidism to all cause and cardiovascular mortality and morbidity in dialysis patients (Palmer et al, JAMA IM, 2011). Phosphate binders have been approved solely based on their ability to lower phosphate concentrations.
There is no evidence that PO4 binders decrease clinically relevant outcomes.
But we know that non-adherence and absenteeism from regular HD, HD inadequacy and dietary indiscretion can lead to hyperphosphatemia in dialysis patients. So, is the relationship between hyperphosphatemia associative or truly causal? If the relationship between hyperphosphatemia and adverse outcomes is truly causal, lowering phosphorus should lead to reduction in mortality and adverse events, but such a definitive trial testing intentional lowering of serum phosphate and its effect of mortality among dialysis patients has never been done.
This thread from Anitha Vijayan provides a nice overview of the entire field in a few tweets
What do the guidelines say?
The most recent- 2017 KDIGO Clinical Practice Guidelines on CKD-MBD suggest lowering elevated phosphate levels toward the normal range which is a 2C recommendation, meaning that it is a recommendation based on low quality of evidence. We attempt to achieve this presently with dietary phosphate restriction, dialysis intensification, and with phosphate binders. Both dietary phosphate restriction and phosphate binders pose significant challenges for dialysis patients and increase the pill burden without evidence to improved patient outcomes
Are there any trials then to support specific phosphorus targets?
Pathway towards knowledge generation in medicine moves from the basic science experiments to observational studies to generate hypotheses and then to appropriately designed RCTs which lead to generation of clinical practice guidelines. But was this pathway followed in terms of phosphate targets or use of phosphate binders? Dr. Ron Wald asks this question in his excellent presentation on this topic (video here). He calls it the “The Great Phosphate Bypass”.
There is no data from an actual RCT with regard to safe phosphate level thresholds or hard endpoints (i.e., mortality, cardiovascular events, and progression of CKD) from treating patients toward different phosphate targets
Why would one want to target a higher phosphorus level?
Lower target phosphate levels in dialysis patients is achieved by dietary phosphate restrictions, phosphate binders and intensification of dialysis- all three interventions put significant burdens on our patients and do not always align with patient priorities and the outcomes which patients care about (Manns et al, cJASN 2014). Phosphate is found in many protein containing foods and restricting these products may not be in the patient's best interest and also has not shown to lead to better outcomes. Phosphate binders increase the pill burden on dialysis patients already dealing with the polypharmacy issue and also escalates costs in dialysis patients - both in terms of dollars and time spent by physicians, pharmacists and dieticians in managing phosphate and phosphate binders. We spend so much blood, toil, and sweat on phosphate control, without knowing if it really helps patients.
And so, going easy on the lower phosphate targets and targeting higher targets may significantly change quality of life and cut the costs for dialysis patients.
Do we have any preliminary data in this area?
Yes we do!
TARGET was a pilot trial, done to test the feasibility of conducting a RCT for intensive vs liberalized phosphate control among hemodialysis recipients (Wald et al, cJASN 2017).
In the intensive arm, phosphate control to normal level (less than 1.5 mmol/L or 4.6 mg/dl) as recommended in the current KDIGO guidelines was achieved by titrating calcium carbonate to a maximum of 3g per day. In the liberalized arm, only rescue use of phosphate binders was allowed if the phosphate level rose to an “alarm” level of more than 2.5 mmol/L or 7.7 mg/dl.
104 patients were randomized: 53 in intensive arm and 51 in control arm, none lost to follow up.
The median phosphate in liberal arm was 1.9 mmol/L (5.9 mg/dl) and 1.5 mmol/L (4.6 mg/dl) in the intensive control arm. This trial demonstrated a meaningful separation between serum phosphate in patients randomized to intensive vs liberalized control, and was done over a period of 26 weeks. One of the notable findings was that in the liberalized arm, the binder dose dropped to zero for many patients, as shown below.
Figure from Wald et al, cJASN 2017
Which brings us to the PHOSPHATE trial.
The PHOSPHATE trial
The aim of this trial (registration link) is to test the hypothesis that compared to a liberal serum phosphate concentration target, intensive lowering of serum phosphate towards the normal level (≤1.50 mmol/L or < 4.65 mg/dl) with phosphate binders reduces the risk of fatal or nonfatal major cardiovascular events in ESKD patients receiving dialysis and improved quality of life in dialysis patients.
In this pragmatic, multinational, randomised, controlled, large, simple trial, a total of 3600 adult ESKD patients receiving dialysis will be randomised either to intensive (≤1.50 mmol/L ie < 4.65 mg/dl) or liberalized (2.0-2.5 mmol/L ie 6.2 to 7.75 mg/dl) serum phosphate target.
What makes PHOSPHATE pragmatic?
The choice and dose of phosphate binders will be at the treating physician's discretion and local practice to achieve and maintain serum phosphate concentration within the required target range according to randomisation.
The inclusion criteria are quite broad and include both HD and PD patients, on dialysis for more than 3 months receiving at least one or more phosphate binders.
The primary endpoint is the composite endpoint of cardiovascular death, non-fatal major cardiovascular or peripheral arterial events. The secondary outcome measures will be individual components of the primary composite endpoint, all-cause death, and utility-based quality of life EQ5D-5L.
No additional blood work will be done in any of the patients, only routinely done labs in dialysis units will be used to adjust doses and for follow up.
The trial implementation is by clinicians and not research coordinators as in a traditional RCT and all acceptable phosphate binders are allowed to be used.
Then what is the HiLo trial, and how is it different?
ESRD patients on dialysis have high rates of hospitalization and deaths, primarily driven by cardiovascular disease and we know from basic science and observational data that hyperphosphatemia is associated with increase in arterial calcification and cardiovascular death.
The HiLo trial is a large, pragmatic , multicenter trial with a cluster randomized design which will test the non-inferiority of liberal phosphate control (target phosphate of 6-7 mg/dl) to currently recommended strict control (target of < 5mg/dl) for all cause hospitalization for dialysis patients. The main secondary hypothesis of HiLo trial is that less stringent phosphate control will reduce risk of all cause mortality, enhance markers of diet and nutrition and improve quality of life.
Unlike the PHOSPHATE trial, the HiLo trial (see Edmonston et al, AJKD 2020) is a cluster RCT in which dialysis units as a whole, and not individual patients will be randomized to intensive or liberal phosphate control. The primary outcome measured in the PHOSPHATE trial is cardiovascular death or non-fatal major cardiovascular event whereas in the HiLo trial, the primary outcome measured is all cause hospitalization rate considered to be a marker of poor health and increased all cause mortality in patients on dialysis.
Watch out for a longer and in-depth discussion of cluster RCTs at a future #NephTrials session.
When will we finally know how to manage phosphorus in dialysis?
Hopefully, with the conclusion of PHOSPHATE and HiLo trials, we will have the answer to whether liberal phosphate control is in fact related to increased cardiovascular outcomes and its effect of quality of life and patient satisfaction with lowered pill burden and liberalized diet restrictions.
Manasi Bapat
East Bay Nephrology Medical Group, Berkeley CA, USA.
Nephrology Social Media Collective faculty
Additional Suggested Reading:
Schwartz et al J Chr Dis 1967; Original description of pragmatic and explanatory trials
Thorpe et al, J Clin Epi 2009; PRECIS tool (original version)
Loudon et al, BMJ 2015; PRECIS-2 publication
PRECIS-2 website
deBoer et al, JASN 2016; ‘Pragmatic Clinical Trials in CKD: Opportunities and Challenges’
Dember et al, JASN 2016; ‘Pragmatic Trials in Maintenance Dialysis: Perspectives from the Kidney Health Initiative’